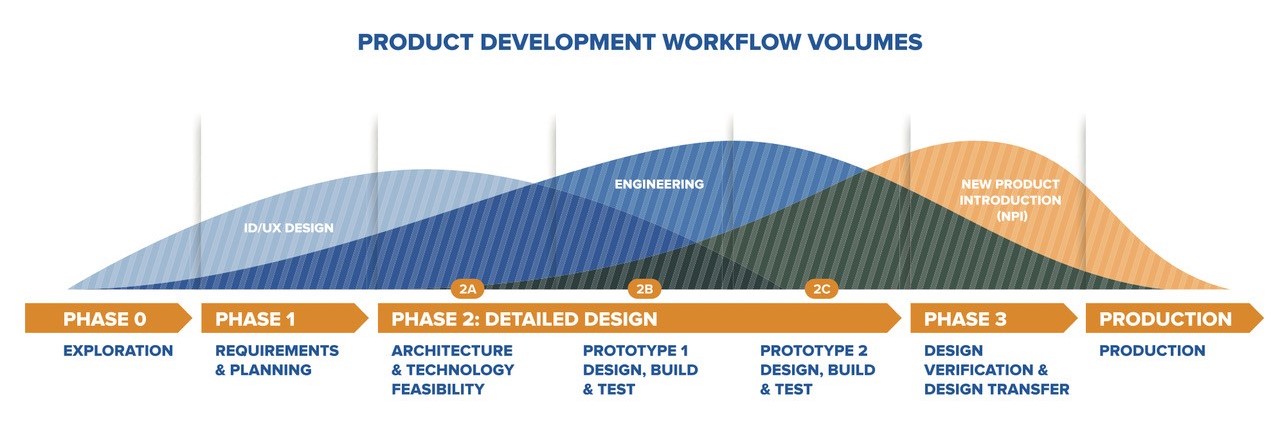
NPI incorporates planning, engineering collaboration, product verification, and manufacturing readiness into a coordinated new product development plan. NPI activities and deliverables are listed below in Phase order according to typical design engineering workflow shown above.
TYPICAL NEW PRODUCT INTRODUCTION (NPI) DELIVERABLES INCLUDE:
PHASE 1: REQUIREMENTS AND PLANNING
- Manufacturing strategy requirements document
PHASE 2A: ARCHITECTURE AND TECHNOLOGY FEASABILITY
- Manufacturing processes review
- Product requirements review
- Critical Performance Metrics (CPM) specifications review
- Project build plan
- Manufacturing plan
- Supply chain strategy development
PHASE 2B: PROTOTYPE 1 – DESIGN, BUILD AND TEST
- ADD Cost of Goods Sold (COGS) worksheet
- RACI chart
- Subassembly strategy development
- Process flow documents
- Test plans and reports
- DFMEA analysis
- Tolerance analysis
- Vendor selection
- Contract manufacturer quote package and selection
- Tooling schedule
- DfX reports (for manufacturability, assembly, serviceability)
PHASE 2C: PROTOTYPE 2 - DESIGN, BUILD AND TEST
- FAI and first article reports
- Quality assurance plan
- Design Transfer package preparation
- Work instructions
- Build status reports
- Build unit tracking
- Assembly fixture and QC tool development
- Contract manufacturer onboarding
- Manufacturing release package delivery
PHASE 3: DESIGN VERIFICATION AND DESIGN TRANSFER
- Root Cause Analysis (RCA)/Defect resolution
- Vendor Management
- Unit build tracking
- Quality metrics verification
- Process validation
- Reliability testing
- Regulatory certification
PRODUCTION
- Manufacturing guidance and ongoing engineering support
- Ongoing quality metrics monitoring and optimization
NEW PRODUCT INTRODUCTION PHASES:
Contact Simplexity to discuss how our New Product Introduction (NPI) team can help you get your next innovation to market.